Efficacy
XERMELO is a tryptophan hydroxylase inhibitor indicated for the treatment of carcinoid syndrome diarrhea in combination with somatostatin analog (SSA) therapy in adults inadequately controlled by SSA therapy.
XERMELO + SSA resulted in sustained bowel movement (BM) reductions over time1
TELESTAR was a 12-week, double-blind, placebo-controlled clinical trial in patients with metastatic neuroendocrine tumors (NETs) and CSD. Patients reported between 4 to 12 BMs daily, despite the use of SSA therapy at a stable dose for at least 3 months.1
Patients were randomly assigned to receive telotristat ethyl 250 mg three times per day + SSA or SSA + placebo (PBO) three times per day.1
In TELESTAR, XERMELO + SSA delivered superior reductions in average daily BM frequency vs SSA + PBO over 12 weeks (primary endpoint; P<0.001).1
Sustained BM reductions continued over 12 weeks
Hr Line


GREATER REDUCTION
IN AVERAGE DAILY BM FREQUENCY VS SSA + PBO: -1.4 VS -0.6 (P<0.001), RESPECTIVELY1
- Allow XERMELO 12 weeks to work, although some patients will see a response within 3 weeks1
- Average daily use of rescue short-acting SSA injections with XERMELO + SSA vs SSA + PBO was 0.3 vs 0.7, respectively1
XERMELO + SSA reduced serotonin as measured by u5-HIAA1,2
At Week 12, u5-HIAA levels decreased with XERMELO + SSA but increased with SSA + PBO (secondary endpoint; P<0.001).1,2


u5-HIAA=urinary 5-hydroxyindoleacetic acid.
There were no significant differences between treatment groups for secondary endpoints of flushing and abdominal pain.2
Durable response with XERMELO + SSA was evaluated2
Secondary endpoint: Durable response defined as ≥30% reduction in BM frequency compared to baseline for ≥50% of the double-blind study period.2


There were no significant differences between treatment groups for secondary endpoints of flushing and abdominal pain.2
XERMELO mechanism of action
See how XERMELO and SSAs work together to address the underlying CSD pathology.1,3

Why is a 12-week trial of XERMELO so important?
To help set evidence-based expectations, explain that it may take up to 12 weeks to see average weekly reductions in BM frequency; however, some patients may see improvement in 3 weeks.1

In a prespecified, exploratory analysis, investigators evaluated the time it took patients to consistently respond to treatment (sustained response). The time to achieve a sustained response was measured from the first dose of the treatment period until the first day of two consecutive weeks that patients experienced a ≥30% reduction in BM frequency.4
The percentage of patients who met this definition of sustained response increased from 4 weeks to 12 weeks.4

75%
Experienced Sustained Reduction IN BM Frequency4
Patients taking XERMELO reported satisfaction levels in an online survey after 6 months of treatment5
Observational analyses are NOT intended for direct comparison with clinical trials and may introduce bias. Real-world evidence can complement data from randomized controlled trials.
In this prospective, observational, open-label, real-world study, patients reported their satisfaction with CSD symptoms via online surveys conducted at baseline and every 6 months up to a maximum of 3 years. 223 patients completed the baseline survey, and 125 patients (56%) completed the 6-month follow-up survey.5
6-month results among patients taking XERMELO included: somewhat to very satisfied (78%), neither satisfied nor dissatisfied (10%), somewhat dissatisfied (9%), and very dissatisfied (4%). The survey results may exhibit a bias, given that only those patients who remained on XERMELO for a duration of 6 months qualified for inclusion in the analysis.5
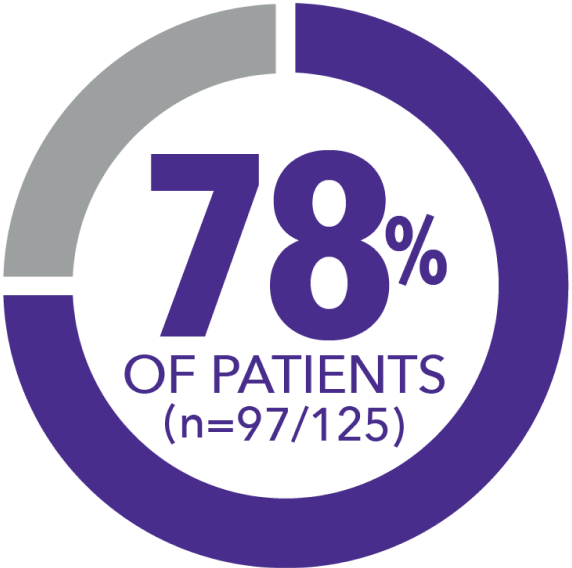
RELAX REGISTRY TRIAL5
Felt "somewhat" to "very satisfied" with control of their CSD symptoms after 6 months of telotristat ethyl treatment
Clinical guidelines recommend XERMELO for patients with uncontrolled CSD6

North American Neuroendocrine Tumor Society (NANETS) guidelines recommend telotristat ethyl (XERMELO) as the appropriate drug of choice, in combination with SSA, for patients with stable radiographic disease and refractory carcinoid syndrome characterized by suboptimal control of diarrhea. Under these circumstances, XERMELO was considered a more appropriate choice than an increase in SSA dose, use of short-acting octreotide, or use of nonspecific antidiarrheal or antitumor therapy.
In situations where there is uncontrolled diarrhea toward the end of the month/4 weeks/dosing cycle, other recommendations may apply.
Patient access and support
Hr lineWays to help patients with CSD on their treatment journey.
About CSD
Hr lineFind questions about refractory CSD symptoms to ask your patients already on SSA therapy.
References:
- XERMELO® (telotristat ethyl). Prescribing Information. TerSera Therapeutics LLC.
- Kulke MH, Hörsch D, Caplin ME, et al. Telotristat ethyl, a tryptophan hydroxylase inhibitor for the treatment of carcinoid syndrome. J Clin Oncol. 2017;35(1):14-23.
- Molina-Cerrillo J, Alonso-Gordoa T, Martínez-Sáez O, Grande E. Inhibition of peripheral synthesis of serotonin as a new target in neuroendocrine tumors. Oncologist. 2016;21(6):701-707.
- Dillon JS, Kulke MH, Hörsch D, et al. Time to sustained improvement in bowel movement frequency with telotristat ethyl: analyses of phase III studies in carcinoid syndrome. J Gastrointest Cancer. 2021;52(1):212-221.
- Li D, Darden C, Osman N, et al. Real-world clinical and patient-reported outcomes from the longitudinal telotristat ethyl treatment registry of patients with neuroendocrine tumors. Cancer Manag Res. 2022;14:3009-3020.
- Strosberg JR, Halfdanarson TR, Bellizzi AM, et al. The North American Neuroendocrine Tumor Society consensus guidelines for surveillance and medical management of midgut neuroendocrine tumors. Pancreas. 2017;46(6):707-714.